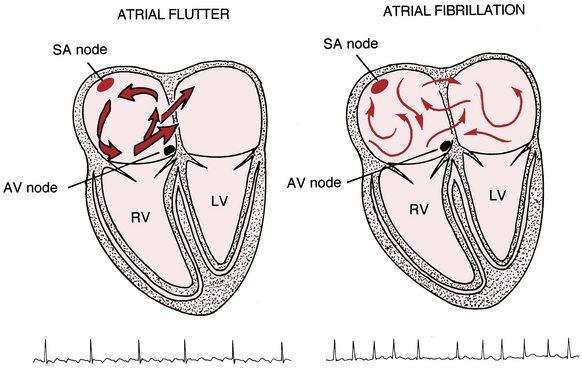
In PH, enlargement of the right atrium (RA) is thought to reflect advancing disease and potential progression to right heart failure as elevated pulmonary and right ventricular pressures are transmitted to the RA. The altered structural and electrophysiological properties of the left atrium in the setting of AF and left heart disease have been well described. Pathophysiology of Atrial Arrhythmias in the Setting of Elevated Right Heart Pressures In a recent study using 24-hour Holter monitoring in 36 patients with PAH or CTEPH, paroxysmal AF was detected in nearly one in three patients, suggesting that the true prevalence of asymptomatic arrhythmia may be much higher. There are no studies using long-term, continuous rhythm surveillance specifically in patients with PH, which would likely increase detection rates of atrial arrhythmias. Age is an important independent risk factor for atrial arrhythmias.Įxisting analyses have relied on intermittent surveillance during clinic visits and hospitalisations due to decompensation or other PH-related issues to detect incident atrial arrhythmias. study, the patients in the Olsson study were generally older (mean 55 years versus 39 years) and had more advanced disease (79 % versus 65 % with WHO functional class III or worse). The variation in reported incidence of atrial arrhythmias in patients with PH may reflect differences in the risk profiles of the patient populations studied. The overall lifetime risk of developing AF or atrial flutter in the absence of heart failure or MI is estimated at approximately 16 % based on Framingham data. By comparison, the incidence of AF or atrial flutter in the general population based on the Rotterdam study was 0.1 % per year among people aged 55–60 years, rising to 2.7 % per year among those aged 80–85 years. studied 280 patients with idiopathic PAH and found a cumulative incidence of 15.8 % over 6 years. This study found a cumulative atrial arrhythmia incidence of 25.1 % over 5 years of follow-up. 
followed 239 patients with idiopathic PAH (n=157) or CTEPH (n=82) and restricted their analysis to AF and atrial flutter. More recent prospective studies have confirmed a relatively high incidence of atrial arrhythmias in PH patients and have attempted to characterise the clinical features of those patients at risk of developing such arrhythmias. A similar series of 72 patients from an Italian center reported a 22 % incidence of atrial arrhythmias over 35 months however, this cohort was also heterogeneous and included patients with WHO group III disease (n=12). The annual incidence of atrial arrhythmias in this cohort was 2.8 % per year, with a cumulative incidence of 11.7 % by the end of the 6-year study (12 patients with atrial flutter, 12 with AF and three with atrioventricular nodal reentrant tachycardia). The patient population was heterogeneous and included patients with PH due to congenital heart disease or inoperable chronic thromboembolic disease (WHO group IV). 
analysed 231 patients undergoing routine surface electrocardiograms as part of their regular outpatient clinic follow-up. The first major observational study by Tongers et al. Most of the early data regarding the burden of sustained atrial arrhythmias in patients with PH were derived from retrospective single- centre analyses. Sarcoidosis, histiocytosis X, lymphangiomatosis, compression of pulmonary vessels (adenopathy, tumour, fibrosing mediastinitis)Įpidemiology of Atrial Arrhythmias in Pulmonary Hypertension Non-thrombotic pulmonary embolism (tumour, parasites, foreign material) Thromboembolic obstruction of distal pulmonary arteriesĤ.3. Thromboembolic obstruction of proximal pulmonary arteriesĤ.2. Pulmonary hypertension due to chronic thrombotic and/or embolic diseaseĤ.1. Pulmonary hypertension associated with lung diseases and/or hypoxemiaģ.1. Left-sided atrial or ventricular heart disease Pulmonary hypertension with left heart diseaseĢ.1. Persistent pulmonary hypertension of the newborn Associated with significant venous or capillary involvementġ.4.2. Other (thyroid disorders, glycogen storage disease, Gaucher’s disease, hereditary haemorrhagic telangiectasia, haemoglobinopathies, chronic myeloproliferative disorders, splenectomy)ġ.4. Congenital systemic-to-pulmonary shuntsġ.3.6.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
